

| Product Name | Fucoidan |
| CAS Number | 9072-19-9 |
| Source | Undaria pinnatifida (Brown seaweed / Wakame) / Fucus vesiculosus (Bladderwrack) |
| Appearance | Light yellow to off-white fine powder |
| Purity (Fucoidan content) | ≥ 85% (standardized) |
| Sulfate content | 15–30% |
| Fucose content | 40–60% |
| Loss on Drying | ≤ 8.0% |
| Heavy Metals | Pb ≤ 2 ppm; As ≤ 2 ppm; Cd ≤ 1 ppm; Hg ≤ 0.1 ppm |
| Storage | Cool, dry, sealed, protected from light |
| Shelf Life | 24 months |
| Package | 1kg, 5kg, 10kg, 25kg |
Need help? Contact our team
Fucoidan is a sulfated polysaccharide found naturally in the cell walls of brown seaweeds (Phaeophyceae), including species such as Undaria pinnatifida (Wakame), Fucus vesiculosus (Bladderwrack), and Laminaria japonica (Kombu). It is composed primarily of L-fucose (a deoxyhexose sugar) combined with sulfate ester groups, along with minor amounts of other monosaccharides (galactose, mannose, xylose, glucuronic acid).
Fucoidan has gained significant attention in nutraceutical, functional food, and cosmetic industries due to its multi-target biological activities, natural origin, and strong safety profile. The global fucoidan market is projected to grow at a CAGR of 10–12% through 2030, driven by immune health and gut health trends.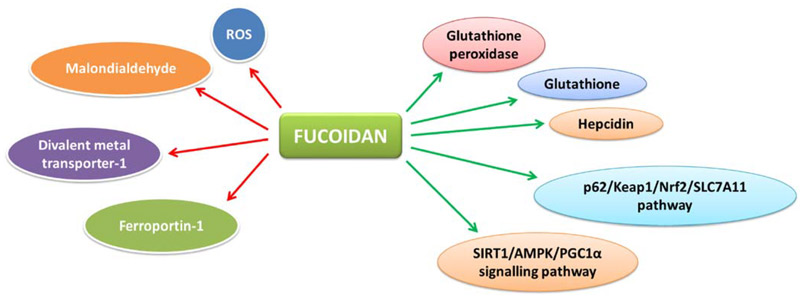
Primary sources comparison:
| Source | Common Name | Fucoidan Content | Sulfate Content | Key Characteristics |
|---|---|---|---|---|
| Undaria pinnatifida | Wakame (Seaweed) | 5–15% dry weight | 15–25% | Most studied, high bioactivity |
| Fucus vesiculosus | Bladderwrack | 10–20% dry weight | 20–30% | High sulfate content |
| Laminaria japonica | Kombu | 2–8% dry weight | 15–25% | Lower cost, large volume |
| Macrocystis pyrifera | Giant kelp | 5–10% dry weight | 15–25% | Sustainable farming |

Wakame
| Application | Typical Dosage | Key Claims |
|---|---|---|
| Daily immune support | 250–500 mg/day | Supports natural immune function |
| Seasonal immune defense | 500–1,000 mg/day (short-term) | Enhanced immune resilience |
| Post-illness recovery | 500–1,000 mg/day | Restores immune balance |
| Application | Typical Dosage | Mechanism |
|---|---|---|
| Gut barrier support | 250–500 mg/day | Reduces intestinal permeability (“leaky gut”) |
| Prebiotic | 250–500 mg/day | Promotes beneficial gut bacteria |
| IBD supportive care | 500–1,000 mg/day | Reduces inflammation (research stage) |
| Application | Typical Dosage | Clinical Evidence |
|---|---|---|
| Joint health / osteoarthritis | 250–500 mg/day | Reduces joint pain and stiffness (preliminary) |
| Anti-inflammatory support | 250–500 mg/day | Reduces systemic inflammation markers |
| Application | Typical Dosage | Research Status |
|---|---|---|
| Cancer supportive care (research) | 500–1,000 mg/day | Preclinical and early human studies |
| Chemotherapy adjunct | 500–1,000 mg/day | Reduces side effects (limited human data) |
note: Anti-cancer claims are not permitted in dietary supplements in any major market. Position as “cellular health support” or “immune health” for commercial products.
| Application | Typical Concentration | Benefits |
|---|---|---|
| Anti-aging creams | 0.5–2% | Hydration, collagen support, antioxidant |
| Skin barrier repair | 0.5–1% | Reduces TEWL, soothes inflammation |
| Anti-acne / soothing | 0.5–1% | Reduces redness, antimicrobial |
LyvBio Co., Ltd. is a supplier of high purity Fucoidan for the nutraceutical, functional food, and cosmetic industries.
Supply capabilities:
Purity: ≥85% fucoidan content (HPLC/colorimetric)
Source: Undaria pinnatifida (Wakame) – standardized
Sulfate content: 15–30% (verified)
Fucose content: 40–60% (verified)
Monthly capacity: 2,000+ kg
Bulk quantities: 100g – 10,000kg
Lead time: 3–5 days (samples), 10–15 days (bulk)
Documentation: COA,UV, HPLC chromatogram, sulfate/fucose analysis, stability data, allergen statement, Non-GMO
| Parameter | Specification | Test Method |
|---|---|---|
| Fucoidan content | ≥ 85% | Colorimetric / UV |
| Sulfate content | 15–30% | Turbidimetric / Ion chromatography |
| Fucose content | 40–60% | HPLC |
| Appearance | Light yellow to off-white powder | Visual |
| Loss on Drying | ≤ 8.0% | USP <731> |
| Ash | ≤ 20% (mineral content from seaweed) | USP <281> |
| pH (1% solution) | 5.0–7.0 | USP <791> |
| Viscosity (1% solution, 25°C) | 50–500 cP | Brookfield |
| Molecular weight distribution | 20,000–200,000 Da | SEC/GPC |
| Heavy Metals | Pb ≤ 2 ppm; As ≤ 2 ppm; Cd ≤ 1 ppm; Hg ≤ 0.1 ppm | ICP-MS |
| Arsenic (inorganic) | ≤ 1 ppm | HPLC-ICP-MS |
| Iodine | ≤ 50 ppm | Titration / IC |
| Microbial | TPC ≤ 1,000 cfu/g; Yeast & Mold ≤ 100 cfu/g; E. coli & Salmonella negative | USP <61> |
Q: What is the difference between fucoidan from different seaweed sources?
A: Undaria pinnatifida (Wakame) is the most clinically studied and widely accepted for nutraceuticals. Fucus vesiculosus (Bladderwrack) has higher sulfate content but also higher iodine. LyvBio uses Undaria pinnatifida for consistent quality and lower iodine.
Q: Is fucoidan vegan?
A: Yes. Fucoidan is derived from brown seaweed (marine algae), not from animals. All LyvBio fucoidan is 100% plant-based.
Q: Does fucoidan have blood-thinning effects?
A: Fucoidan has anti-coagulant activity in vitro (similar to heparin but much weaker). At typical supplement doses (250–1,000 mg), clinically significant blood thinning is unlikely. However, caution is advised for individuals on anticoagulant medications.
Q: What is the difference between high MW and low MW fucoidan?
A: High MW (>100k Da) has lower absorption but may be more effective for gut-local effects. Low MW (20–50k Da) has higher systemic bioavailability for immune modulation. Oligo-fucoidan (<10k Da) has the highest absorption for premium formulations.
Q: Is fucoidan stable in gummy formulations?
A: Yes, with proper formulation. Use low MW or oligo-fucoidan for better solubility. Taste masking (fruit flavors) is essential to cover the mild seaweed taste.
Q: What is the iodine content of your fucoidan?
A: LyvBio fucoidan is extracted and purified to remove most iodine. Typical iodine content is ≤50 ppm (significantly lower than whole seaweed, which can contain 500–5,000 ppm). This is safe for daily use.
Q: What is your MOQ?
A: Sample orders: 100g–1kg. Trial production: 5–25 kg. Commercial: Negotiable (100kg+ receives volume pricing). Lead time: 3–5 days (samples); 10–15 days (bulk).
📧 Email: info@lyvbio.com
🌐 Website: https://www.lyvbio.com